SOUTH SAN FRANCISCO, Calif. — Senti Biosciences Holdings, Inc. (Nasdaq: SNTI) (“Senti Bio” or the “Company”), a clinical-stage biotechnology company developing next-generation cell and gene therapies using its proprietary Gene Circuit platform, today announced the successful completion of a Type B Initial Comprehensive Multidisciplinary Regenerative Medicine Advanced Therapy (RMAT) meeting with the U.S. Food and Drug Administration (FDA) regarding SENTI-202, the Company’s first-in-class Logic Gated off-the-shelf CAR-NK cell therapy for relapsed/refractory acute myeloid leukemia (R/R AML) and updated Phase 1 clinical data.
Following the RMAT meeting, the Company has finalized its pivotal clinical and chemistry, manufacturing and controls (CMC) strategy for SENTI-202. The Company plans to implement a single-arm, multi-center pivotal trial intended to support potential SENTI-202 registration in patients with R/R AML. This study is expected to evaluate SENTI-202 administered following lymphodepletion (LD) chemotherapy in a patient population consistent with the Phase 1 trial population.
In addition to the positive RMAT meeting, after conducting exploratory efficacy covariate analysis of the Phase 1 trial results, Senti has identified a specific Donor X attribute that correlates with efficacy of SENTI-202, with 50% (7/14) of the patients achieving a cCR when they received any SENTI-202 doses manufactured from Donor X-characteristic-derived NK cells in Cycle 1 versus 12.5% (1/8) achieving a cCR when they received SENTI-202 manufactured from non-Donor X NK cells (see Table below). As a result of this discovery, all future SENTI-202 manufacturing, including for pivotal study use, will use Donor X material. The Donor X attribute is found in ~50% of adult donors, and published literature supports increased NK cell cytotoxicity in donors with this phenotype. The Donor X NK phenotype is independent of HLA or KIR matching, thus supporting SENTI-202’s allogeneic off-the-shelf usage. Retrospective analysis of preclinical MV4-11 NSG mouse model data confirmed increased activity and survival with Donor X product (see Figure below).
Senti Bio also announced that SENTI-202 continues to exhibit durable MRD-negative responses in the full 22 patient Phase 1 trial, which compares favorably with current FDA approved therapies for R/R AML. At RP2D, across all patients receiving a mix of Donor X and non-Donor X material, an ORR of 44% and cCR of 37.5% was observed with 100% of CRs being MRD negative. The complete remissions continue to be durable, with all the CR/CRh responders who were in remission as of the data-cut supporting the oral presentation at the 2025 ASH annual meeting continuing to maintain remission with an additional 7 months of follow up, the longest duration being 21+ months.
“This positive FDA RMAT meeting marks a transformational moment for Senti Bio and significantly advances our path toward potential registration of SENTI-202,” said Tim Lu, M.D., Ph.D., Chief Executive Officer and Co-Founder of Senti Bio. “This news, combined with the compelling clinical responses observed to date that led to refinements in our donor selection strategy, positions us to advance SENTI-202 toward a potential registrational study in relapsed/refractory AML. We believe this milestone further validates both our Gene Circuit platform and the differentiated therapeutic potential of Logic Gated cell therapies.”
FDA previously granted RMAT designation to SENTI-202. This program is intended to facilitate the expedited development and review of regenerative medicine therapies addressing serious or life-threatening diseases.
“The FDA feedback provides important clarity around our registrational development strategy and further supports our conviction in the SENTI-202 program,” said Kanya Rajangam, M.D., Ph.D., Chief Medical Officer of Senti Bio. “The excellent clinical activity observed thus far, including MRD-negative durable complete remissions alongside a favorable safety profile, gives us confidence as we transition toward later-stage development. We are focused on rapidly implementing the pivotal study while also exploring potential expansion opportunities in newly diagnosed AML and pediatric AML. Since the filing of our IND, Senti has focused on donor selection to minimize variability. We are in a strong position as we prepare for our clinical trials with the identification of a donor phenotype that correlates with increased activity and continues to support SENTI-202’s allogeneic manufacturing.”
Relapsed/refractory AML remains an aggressive hematologic malignancy with limited therapeutic options and poor long-term survival outcomes. Senti Bio believes SENTI-202’s differentiated mechanism, off-the-shelf availability, and encouraging early clinical profile position the program as a potentially important next-generation treatment option for AML patients.
| Table: Phase 1 SENTI-202-101 Trial R/R AML Patient Efficacy Data Based on Donor Phenotype |
||
| All Patients (N=22) |
Any Donor X in Cycle 1 | No Donor X in Cycle 1 |
| ORR (Overall Response Rate) | 8/14 (57%) | 2/8 (25%) |
| cCR | 7/14 (50%) | 1/8 (12.5%) |
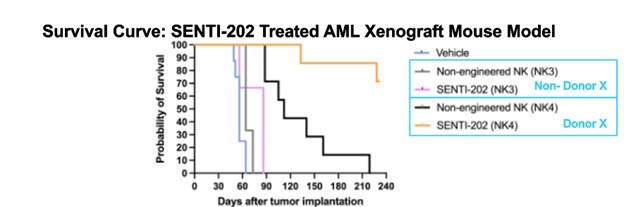
| Vehicle | Non-engineered NK (NK3) |
SENTI-202 (NK3) | Non-engineered NK (NK4) |
SENTI-202 (NK4) | |
| Median Survival (d) | 56.0 | 64.0 | 86.0 | 112.0 | Not Reached |
| Figure: Retrospective analysis of preclinical MV4-11 NSG mouse model data confirms increased activity and survival with SENTI-202 made from Donor X product. Donor X characteristic was confirmed post-hoc. |
|||||
About SENTI-202
SENTI-202 is a first-in-class Logic Gated off-the-shelf CAR-NK cell therapy designed to selectively target and eliminate CD33 and/or FLT3 expressing hematologic malignancies, including AML and myelodysplastic syndrome (MDS), while sparing healthy bone marrow cells. SENTI-202 incorporates multiple engineered Gene Circuits, including OR GATE and NOT GATE logic systems and calibrated-release IL-15, to improve tumor specificity, persistence, and therapeutic activity.
SENTI-202 has received Regenerative Medicine Advanced Therapy (RMAT) designation and Orphan Drug Designation (ODD) from the U.S. Food and Drug Administration.
About the Phase 1 Study
The multinational, multicenter dose-finding study of SENTI-202 (NCT06325748) comprised an initial dose finding using a modified “3+3” study design to determine the maximum tolerated dose (MTD) and/or recommended phase two dose (RP2D) of SENTI-202 when administered after lymphodepleting chemotherapy (Part 1) followed by disease-specific expansion cohorts at the RP2D (Part 2).
The primary objectives were to evaluate safety, determine the MTD and RP2D, and assess efficacy in expansion cohorts using ELN 2022 consensus criteria for AML, with key secondary objectives including measurable residual disease assessment, pharmacokinetics, and pharmacodynamics using CyTOF on serial bone marrow samples. For more information visit clinicaltrials.gov.
About Senti Bio
Senti Bio is a clinical stage biotechnology company developing a new generation of cell and gene therapies for patients living with incurable diseases. To achieve this, Senti Bio is leveraging its synthetic biology platform to engineer Gene Circuits into new medicines with enhanced precision and control. These Gene Circuits are designed to precisely kill cancer cells, to spare healthy cells, to increase specificity to target tissues, and/or to be controllable even after administration. The Company’s wholly-owned pipeline comprises cell therapies engineered with Gene Circuits to target challenging liquid and solid tumor indications. Senti Bio’s Gene Circuits have been shown preclinically to work in both NK and T cells. Senti Bio has also preclinically demonstrated the potential breadth of Gene Circuits in other modalities and diseases outside of oncology, and continues to advance these capabilities through partnerships. To learn more visit https://www.sentibio.com/.
Investor Contact
JTC Team, LLC
Jenene Thomas
(908) 824-0775
[email protected]
