WILMETTE, Ill. — Monopar Therapeutics Inc. (“Monopar” or the “Company”) (Nasdaq: MNPR), a clinical-stage biopharmaceutical company developing innovative treatments for patients with unmet medical needs, today announced that the Journal of Hepatology has published a peer-reviewed Letter to the Editor (link), authored by leading Wilson disease physicians, entitled “Oral Bis-choline Tetrathiomolybdate Rapidly Improves Copper Balance in Patients with Wilson Disease.” Wilson disease is a rare and progressive genetic condition in which the body’s pathway for removing excess copper is compromised, leading to damage from toxic copper build-up in organs such as the liver and brain.
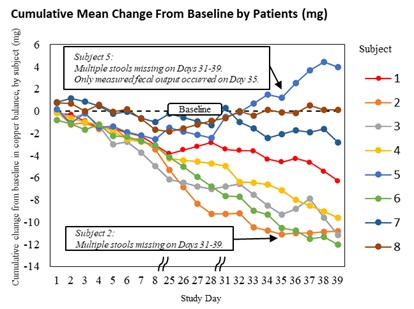
In the Letter to the Editor, the authors explain that the earlier conclusion from the Phase 2 ALXN1840-WD-204 study (NCT04573309) – that ALXN1840 did not promote copper excretion – was based on a methodological limitation in the copper balance equation, which only accounted for certain routes of copper loss. By controlling for the other routes of copper loss by comparing pre- and post-ALXN1840 treatment, the analysis demonstrates that ALXN1840 statistically significantly improved copper balance (increases copper excretion) over the duration of the study. Supplementary materials published alongside the Letter illustrate how the same primary copper balance data produced both the prior sponsor’s analysis as well as the results presented in the Letter.
Key findings on the effect of ALXN1840 on copper balance reported in the Letter:
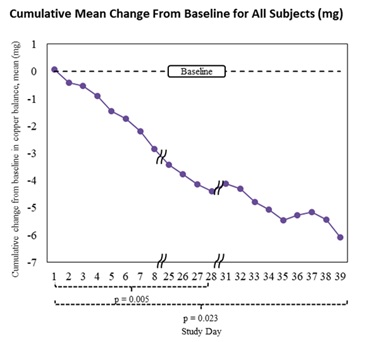
- 15 mg/day treatment period: mean daily difference -0.367 mg (p=0.005)
- Overall treatment period (includes patients with dose changes to 15 mg every other day and to 30 mg/day): mean daily difference -0.289 mg (p=0.023)
- Cumulative mean change from baseline: -6.08 mg (95% CI: -10.18 mg to -1.98 mg), see graphs below
About Monopar Therapeutics Inc.
Monopar Therapeutics is a clinical-stage biopharmaceutical company with late-stage ALXN1840 for Wilson disease, and radiopharmaceutical programs including Phase 1-stage MNPR-101-Zr for imaging advanced cancers, and Phase 1a-stage MNPR-101-Lu and late preclinical-stage MNPR-101-Ac225 for the treatment of advanced cancers. For more information, visit: www.monopartx.com.
Contact
Monopar Therapeutics Inc.
Investor Relations
Quan Vu
Chief Financial Officer
[email protected]
